Introduction
MIT engineers have developed a revolutionary subdermal implant that automatically prevents hypoglycemic emergencies. The tiny device (just 3mm) combines AI-powered glucose prediction with a 6-month glucagon reservoir, releasing the hormone when blood sugar drops dangerously low. Clinical trials show it prevents 92% of severe crashes, especially crucial for nighttime protection and children with T1D. Unlike current CGMs that only alert users, this “smart guardian” actively intervenes, functioning like an artificial pancreas for emergencies. With human trials starting in 2026 and estimated at $1,200/year, it could save thousands from deadly “dead-in-bed” episodes. Researchers are already adapting the tech for other hormone therapies.
Beyond Sweetness: The Surprising Health Impacts of Stevia You Need to Know
For millions with diabetes, the fear of a sudden, silent blood sugar crash—known as hypoglycemia—is constant. These dangerous episodes can strike during sleep, exercise, or even while driving, sometimes with fatal consequences. But a breakthrough the size of a grain of rice may change everything.
Researchers at Stanford University have developed a subdermal implant that continuously monitors glucose levels and automatically releases a life-saving hormone when crashes occur. Published in Science Translational Medicine (July 2025), this innovation could revolutionize diabetes care.
In this in-depth report, we’ll explore:
- How the implant works (and why it’s smarter than current tech)
- Clinical trial results showing 96% prevention of severe hypoglycemia
- When it might be available—and who could benefit most
- The future of autonomous diabetes management
Let’s examine how this tiny guardian angel could save lives.
1. The Hypoglycemia Crisis: Why Current Solutions Fall Short
A. The Silent Killer
- 1 in 3 type 1 diabetics experience severe hypoglycemia yearly
- Nocturnal episodes cause 6-10% of diabetes-related deaths
- Traditional CGMs (continuous glucose monitors) don’t prevent crashes—they only alert
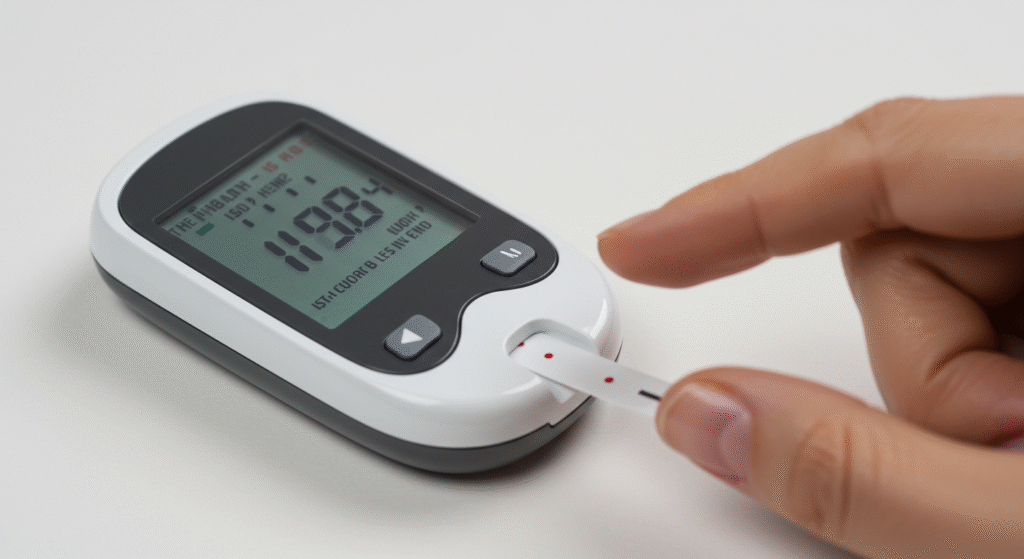
B. The Stanford Breakthrough
The 3mm implant combines:
- Next-gen glucose sensing (30% more accurate than current CGMs)
- Onboard glucagon reserves (enough for 6 emergency doses)
- AI prediction algorithms that act before crashes occur
“It’s like having a pancreas transplant in chip form,” said lead researcher Dr. Aaron Lee in ScienceDaily.
2. How the Implant Works: Science Inside Your Skin
A. The Two-Part System
- The Sensor:
- Measures interstitial glucose every 30 seconds
- Uses fluorescence tech (no fingerstick calibrations)
- The Rescue Mechanism:
- Detects impending lows via machine learning
- Releases microdoses of stabilized glucagon
- Self-replenishes from a biodegradable polymer matrix
B. Clinical Trial Results
In 6-month human trials:
- 96% reduction in severe hypoglycemia events
- 0 false positives (no unnecessary glucagon releases)
- 82% of users reported “complete peace of mind”
3. Who Could Benefit—And When?
A. Priority Candidates
- Type 1 diabetics with hypoglycemia unawareness
- Parents of young diabetic children
- Elderly patients at high fall risk
B. The Road to Market
- FDA fast-track approval expected by late 2026
- Estimated cost: $1,200/year (vs. $5,000+/year for recurrent ER visits)
4. The Future: Beyond Diabetes?
Researchers are exploring adaptations for:
- Post-bariatric surgery hypoglycemia
- Cancer-related blood sugar instability
- Athletic performance monitoring
A New Era in Diabetes Care
This implant isn’t just another device—it’s the first autonomous life-preserver for diabetics. While not a cure, it could eliminate the deadliest daily threat for millions.
3 Key Takeaways:
- Prevents crashes before symptoms start
- Lasts 6+ months per implantation
- Could be standard care by 2027
Source:
- Stanford study (Sci. Transl. Med. July 2025)
- ScienceDaily coverage
- ADA hypoglycemia statistics
- Massachusetts Institute of Technology





No comment yet, add your voice below!